The Two Sessions Signal the “15th Five-Year Plan”, And Eight Leading-Edge Medical Tracks Are Set to Enter a Golden Five Years!
Published By: VB Data
Author: Zhang Xiaoxu
Time: 03/07/2026
It is once again the time for the annual Two Sessions (The National People’s Congress of the People’s Republic of China and the National Committee of the Chinese People’s Political Consultative Conference).
In the opening year of the 15th Five-Year Plan, the 15th Five-Year Plan, which is set to be released in 2026, has attracted widespread attention from all sectors of society, and the review of the draft outline has also become one of the core agendas of Two Sessions.
In order to promote the implementation of the tasks of the 15th Five-Year Plan, the draft outline has proposed 109 major projects in six areas, clarifying key directions for the development of various industries over the next five years.
In the healthcare, the draft outline not only continues to advance routine tasks such as public health, medical services, primary healthcare, elderly care and childbirth, but further addresses medical-related content in “Hard Tech”, including new industries and cutting-edge scientific research, thereby indicating the direction for frontier technological innovation in the medical over the next five years.
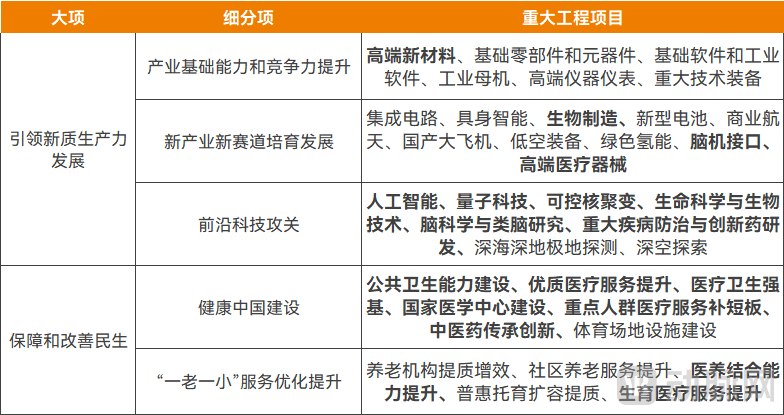
Medical and Health-Related Content in the Draft Outline of the “15th Five-Year Plan”
Source: Xinhua News Agency
First: Brain-Computer Interface (BCI) and Brain Science
BCI are undoubtedly one of the hottest areas in medical health during the Two Sessions.
Following its emphasis by various local Two Sessions earlier this year, BCI have now, for the first time, been included in the government work report of the Two Sessions, listed alongside frontier technologies such as future energy, quantum technology and embodied intelligence as a key industry to be cultivated for the future. More importantly, the draft outline includes BCI among the projects for fostering new industries, and incorporates brain science and brain-inspired research into major projects for tackling cutting-edge scientific challenges.
The popularity of BCI in the industrial sector has already become evident. According to the Brain-Computer Interface Technology and Application Research Report (2025) released by the China Academy of Information and Communications Technology and the Brain-Computer Interface Industry Alliance, the total number of brain-computer interface enterprises in China has exceeded 200. As the main application area, BCI applications in healthcare sectors have received focused attention from innovative companies and investors. According to incomplete statistics from the Dongmaicheng database, in 2025, there were a total of 24 financing events for BCI in the healthcare, representing a year-on-year increase of 30%.
In clinical diagnostic and treatment scenarios such as neurodegenerative diseases and mental illness, BCI can explore pathological and physiological mechanisms, innovate neuromodulation technologies, and effectively improve disease diagnosis and treatment outcomes. For patients with motor, language and sensory impairments, BCI can create new solutions for motor assistance, communication aid and sensory rehabilitation, thereby enhancing the quality of patient recovery. In scenarios related to brain disease prevention and health, BCI products can monitor and analyze brain activity features in real time, prevent potential health issues, and reduce the risk of severe conditions for patients.
According to the planning of relevant national ministries and commissions, in the next five years, China will cultivate 2 to 3 leading enterprises with global influence and a number of specialized and innovative small and medium-sized enterprises, significantly enhancing the innovation capability of the country’s BCI industry. In terms of basic hardware and software research, the focus will be on innovating brain signal sensing components, breaking through key brain-computer chips, and improving software tool platforms. In terms of product, efforts will accelerate breakthroughs in the implantable devices, promote mass production and iteration of non-implantable devices, and widely develop auxiliary equipment.
Driven by top design and industrial innovation, BCI is about to enter five years of rapid development.
Second: Innovative Drug
In the draft outline, major disease prevention and treatment, as well as innovative drug development, have been included as key projects in frontier technology research, providing direction for the development of the industry.
As of now, our country has achieved significant phased results in innovative drugs: according to data from the National Medical Products Administration(NMPA), 76 innovative drugs were approved for market launch in 2025, setting a historical record; in the past five years, a total of 230 innovative drugs have been approved, realizing substantial growth.
Even more substantial achievements are still in reserve. From the perspective of research and development, innovative drugs have entered a new heyday. In 2025, domestic innovative drug pipeline BD transactions were active, and corporate IPOs were frequently realized, greatly boosting industry confidence. The continued boom in BD transactions directly reflects multinational pharmaceutical companies’ (MNCs) high recognition of China’s innovative drug R&D capabilities.
Entering 2026, this momentum continued: in just January, the total value of domestic innovative drug BD transactions reached 34.6 billion USD, with upfront payments exceeding 3 billion USD, making the total transaction value equivalent to one-quarter of the entire year of 2025. This indicates that China’s globalization of innovative drugs is unprecedented pace, with more innovative drugs progressing from clinical stages to practical application under global technological and financial support.
In the future, technologies such as small nucleic acid therapeutics, gene and cell therapies, and small molecule therapeutics, as well as disease areas including oncology, autoimmune, neurology, and metabolism, will be key directions for research and development breakthroughs and capital deployment.
In the policy, efforts are continuously being intensified. China will promote the implementation of a data protection system for pharmaceutical trials and study the establishment of market exclusivity periods for pediatric drugs and drugs for rare diseases. For innovative drugs with new mechanisms and new targets, communication, clinical trials, regulatory submissions, and the full chain of review and approval services will be strengthened to support these drugs in achieving “China-first launch”. At the same time, the review and approval processes for cell and gene therapy products will be optimized to further streamline the pathway for innovative drugs to enter the market.
Third: High-End Medical Devices
In the draft outline, high-end medical devices are one of the major projects for cultivating new industries.
The innovative breakthroughs in high-end medical devices are a long-term and ongoing process. Through the collaborative efforts of various parties in recent years, domestic achievements have been made in some products, and based on a relatively high market share domestically, they have even begun to expand overseas.
In 2025, the NMPA issued the Announcement on Measures to Optimize Full Lifecycle Supervision to Support Innovation and Development of High-end Medical Devices, providing policy-integrated innovation to support the development of the high-end medical device industry. It proposed comprehensive support policies covering the entire chain from research and development project initiation, registration application, review and approval, to post-market re-evaluation. Focusing on core areas, it also formulated the Priority Approval Catalogue for High-end Medical Devices (2025), further supporting high-end medical devices with significant clinical application value and urgent clinical needs.
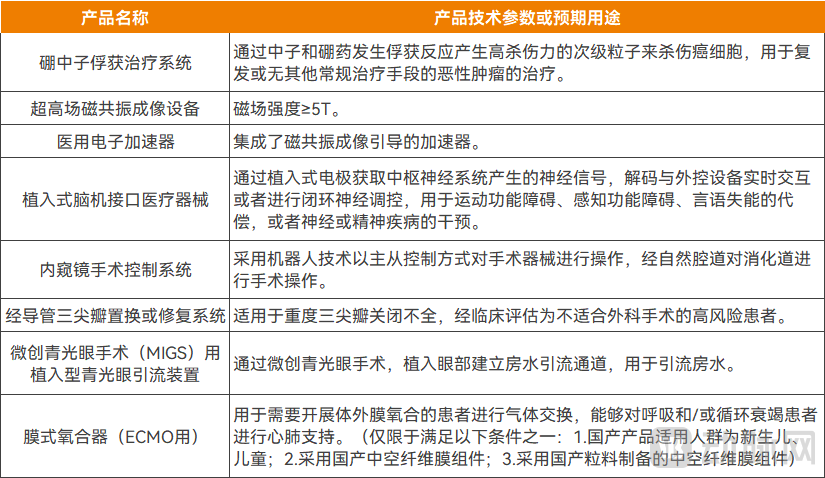
Priority Approval for the Catalog of High-End Medical Devices (2025)
Source: NMPA
In addition, national and local policies have provided strong support for domestically produced high-end medical devices in terms of bidding and procurement, as well as the first set of policies. However, it should not be overlooked that domestic products still have significant shortcomings in certain high-end areas. For example, there is considerable room for advancement in core components and key parts of high-end imaging equipment, complete systems of surgical robots and various other therapeutic devices, as well as high-end interventional consumables. In the future, establishing a multidisciplinary and collaborative mechanism will become an important direction for the industry to achieve key breakthroughs.
Fourth: Biomedical Materials
Among the 109 major projects proposed in the draft outline, high-end new materials are one of the projects aimed at enhancing industrial foundational capabilities and competitiveness.
In medical, biomedical materials are an essential material, supporting the development of the medical device industry. As a part of high-end new materials, they are indispensable.
At present, China is accelerating breakthroughs in biomedical materials. The most typical initiative is the “Biomedical Materials Innovation Task Leading” jointly launched by the Ministry of Industry and Information Technology of People’s Republic of China and the National Medical Products Administration. In 2023 and 2025, two lists were successively released, with dozens of material categories and more than one hundred enterprises and institutions selected.

Key Biomedical Materials and Applications
Source: Ministry of Industry and Information Technology
Focusing on key research breakthroughs is expected to address the long-standing dilemma in biomedical materials, break foreign technological monopolies, and achieve independent and controllable production of critical materials. At the same time, in addition to innovative enterprises in the medical and healthcare sector, this initiative also involves upstream energy companies, universities, and research institutions, promoting collaborative innovation across the entire industry chain. This is conducive to accelerating the tackling of technical challenges in biomedical materials and enhancing the overall competitiveness of the industry chain.
Fifth: Medical AI
AI is one of the key directions for cutting-edge technology research and development in the draft outline of the 15th Five-Year Plan. As an important branch of it, medical AI is experiencing strong growth momentum.
Currently, the domestic medical AI has moved from technical pilot projects into a new stage of large-scale implementation. Especially since 2025, the medical AI has shown two significant changes: the breakthrough evolution of large models and the large-scale participation of medical institutions.
Over the past year, domestic large-scale medical models have experienced explosive growth. With continuous technological breakthroughs, medical large models have transitioned from rule-driven to data-driven approaches, advancing from single-modality to multi-modality integration; their capabilities have also expanded from recognition and generation to include logical reasoning, increasingly approaching the level of human cognition. Meanwhile, public hospitals, represented by the top 100 domestic hospitals, have extensively deployed AI infrastructures and various applications driven by the surge of large models.
According to the Dongmai Think Tank’s 2025 Medical Artificial Intelligence Report, at the current stage, medical AI has gained multiple benefits from capital, policy, and collaboration with doctors. However, in order to transform these resources into sustainable and realizable value, it is still necessary to further reduce research and development costs, resolve the value divergence of medical AI (the difficulty in synchronizing patient experience with hospital benefits), and achieve substantial progress in business models and payment systems.
With the acceleration of AI and the broadening accessibility of its applications, future competition in medical AI will no longer be confined to the technology itself. It will increasingly require the development of a value system around AI products that benefits multiple stakeholders. For medical institutions, only by transforming medical AI from a mere cost input into an asset that enhances core competitiveness can the long-term value of AI be fully realized.
Sixth: Biomanufacturing
In the draft outline, biomanufacturing is regarded as one of the major projects for cultivating new industries.
As an important technological supporting the bioeconomy, biomanufacturing is continuously evolving toward high-throughput, intelligent and controllable directions. However, biomanufacturing still faces numerous challenges. Among them, pilot-scale testing is the critical bridge that connects laboratory samples to market products and serves as the core support for scientific and technological achievements.
At present, the Ministry of Industry and Information Technology and the National Development and Reform Commission are vigorously promoting the enhancement of pilot-test in biomanufacturing and establishing pilot-test construction platforms. In November 2025, the first batch of platform lists was announced. In the biopharmaceuticals, this includes enterprises such as JOINN Biologics, Bloomage Biotech, Changchun Biologics, Qilu Pharmaceutical, Chutian Science and Technology, Womei Biology, as well as research institutions such as the Frontier Research Institute of Synthetic Biology at Tianjin University, the Wenzhou Institute UCAS, and the Xiang An Biomedicine Laboratory.
With the systematic layout of pilot-test capacity building platforms, as well as the iterative application of AI technology and other technologies in biomanufacturing, the biomanufacturing is expected to achieve substantial breakthroughs in this critical segment over the next five years, thereby driving the overall better development of the industry.
Seventh: Life Sciences and Biotechnology
ciences and biotechnology have been included as major projects for key technological breakthroughs.
In recent years, our country has rapidly developed in frontier fields such as gene editing, synthetic biology, stem cells and regenerative medicine, brain science, as well as the cross-integration of large biological models and artificial intelligence, resulting in a continuous emergence of new technologies and innovative therapies.
The continuous development of life omics technologies is facilitating panoramic research in life sciences; cutting-edge molecular diagnostics are continually innovating, aiding in early disease detection and screening; gene therapy is steadily advancing in more genetic and rare diseases; multiple breakthroughs in regenerative medicine are accelerating clinical translation; antibody drug technology has reached a relatively mature stage, with new types of antibody drugs gradually moving from clinical trials to application; innovations in targeted protein degradation are breaking the limitations of previously undruggable protein targets. From a series of significant advancements outlined in the China National Center for Biotechnology Development’s 2025 China Life Sciences and Biotechnology Development Report, one can clearly perceive the remarkable progress in China’s life sciences and biotechnology.
It is foreseeable that, driven by multiple forces such as technology, policy and capital, more fundamental research achievements in life sciences and biotechnology will accelerate their transformation into practical products, providing a firmer support for innovative drugs and innovative medical devices.
Eighth: Medical Applications of Other Cutting-Edge Technologies
The draft outline and the government work report identify several other frontier technologies as key areas for research or development. Among them, quantum technology, controlled nuclear fusion, and green hydrogen energy can all be applied to the medical, playing a critical role in disease diagnosis, drug research and development, and other processes.
For example, in quantum technology, X-Magtech Technology integrates quantum technology into the medical industry to meet the needs of diagnosing major cardiovascular and cerebrovascular diseases and scientific research. It has independently developed two high-end innovative medical devices: a magnetocardiograph and a magnetoencephalograph. Boson Quantum collaborates with research institutions and medical organizations to explore practical quantum computing applications in scenarios such as protein structure prediction, molecular similarity screening, peptide docking and allosteric site prediction; it also shares quantum computing power and data resources to advance the process of drug development and clinical validation.
In controlled nuclear fusion, regions such as Sichuan and Anhui have already issued policies, making the commercialization of controlled nuclear fusion one of their key future industries, with medicine being one of the application scenarios. Taking Zhongke Medical Valley in Hefei as an example, in January 2026, it proposed the “Fusion Medicine” strategy, with the first batch of projects focusing on research and development breakthroughs in applications of radiation-resistant microorganisms in medical, new advances in FLASH radiotherapy and low-dose treatments, and the latest developments in BNCT systems.
Finally
In cutting-edge technology, from top-level design and basic research to the transformation of results, as well as industrial implementation and practical application, it is still necessary to build a complete chain and undergo a relatively long cycle. The path of industrial development is lengthy and cannot be achieved without deep coordination and close cooperation among all parties in the industrial chain.
Under the guidance of national policies, we also look forward to research institutions, innovative enterprises and investment organizations working together, jointly contributing to achieving globally leading innovative breakthroughs in the healthcare industry.